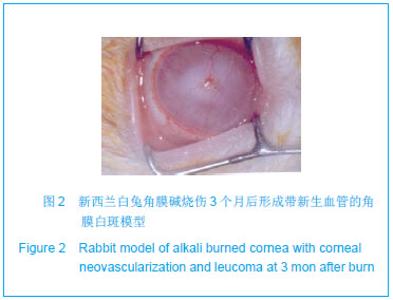
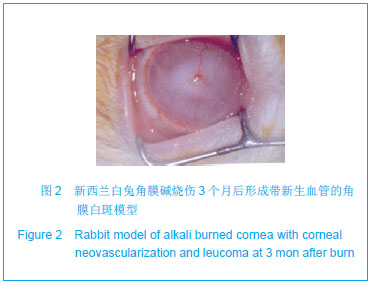
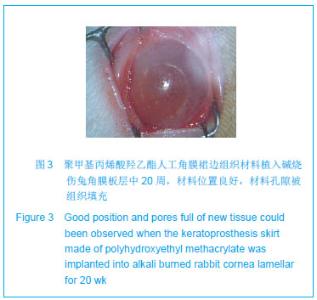
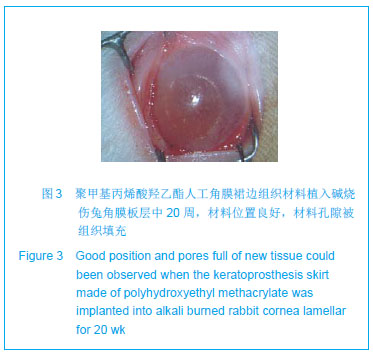
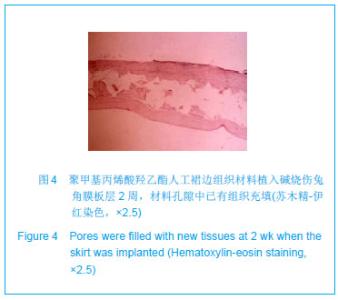
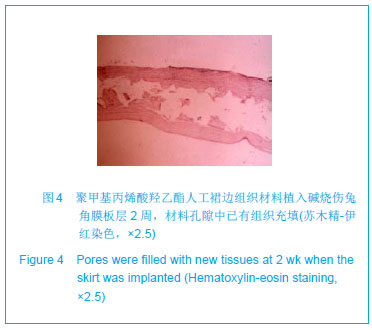
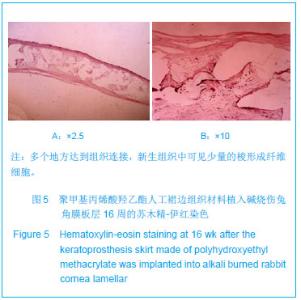
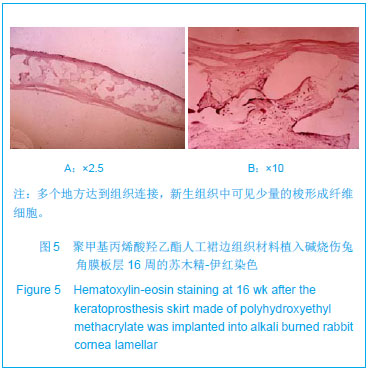
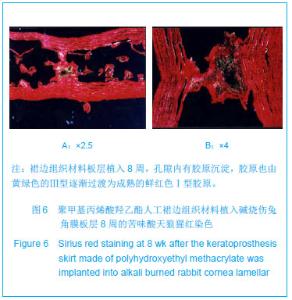
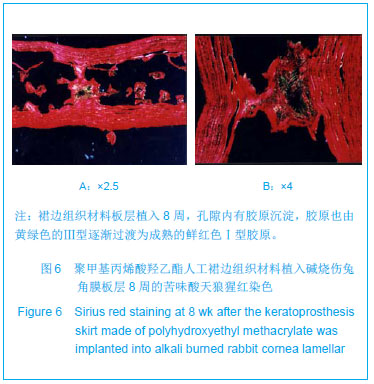
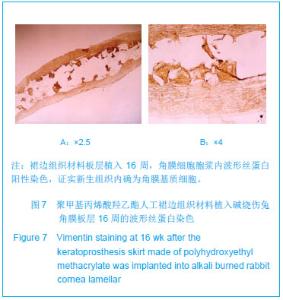
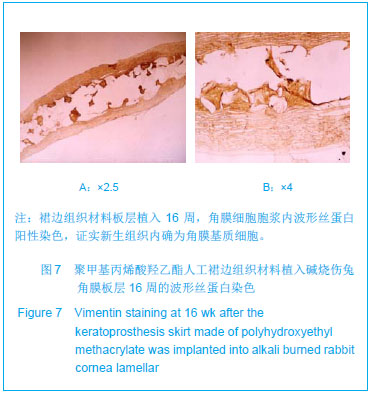
| [1]蒙艳春,高峰,黄一飞,等.兔角膜植入改性HEMA人工角膜支架材料的实验研究[J].军医进修学院学报,2002,23(2):150-152.[2]胡平,高峰,许波.一种用本体聚合粒子滤出法制备人工角膜的方法.中国,发明专利,公开号:CN1318351A[3]Gao F,Meng YC,Qu B,et al.Preparation of artifical cornea with a central optic region and porous skirt.Chin J Biomed Eng. 2002;(10):7-9.[4]胡平,许波,高峰.一种用溶胀聚合粒子滤出法制备人工角膜的方法.中国,发明专利,公开号:CN01129695.X[5]Kirkham SM,Dangel ME.The keratopsthesis: improved biocompatibility through design and surface modification. Ophthalmic Surg.1991;22(8):455-461.[6]Pintucci S,Pintucci F,Cecconi M,et al.New Dacron tissue colonisable keratoprosthesis: clinical experimence.Br J Ophthalmol.1995;79(9):825-829. [7]Kim HC.The experimental Seoul-type keratoprosthesis. Korean J Ophth almol. 1992;6(2):55-61.[8]Dohlman CH,Terada H.Keratoprosthesis in pemphigoid and Steven- Johnson sydrome.Adv Exp Med Biol.1998;438: 1021-1025.[9]Wapner FJ,Srinivasan BD.Calcific band keratopathy on a keratoprosthesis.Cornea.1993;12(1):72-73.[10]Claes C,Worst J.Retinal detachment surgery following implantation of a kerato- prosthesis.Bull Soc Belge Ophthalmol.1992;243:167-169. [11]Hile K,Landau H,Ruprecht KW.Improvement of the osteo-odonto-keratoprosthesis according to Strampelli:influence of diameter of PMMA cylinder on visual field. Graefes Arch Clin Exp Ophthalmol.1999;237:308-312.[12]Griffith M,Osborne R,Munger R,et al.Functional human corneal equivalents constructed from cell lines.Science. 1999; 286(5447):2169-2172.[13]梁丹, 陈家祺, 李永平,等.角膜板层植入两种不同孔径膨体聚四氟乙烯的实验研究[J].眼科学报,1999,15(4):246-249.[14]Patel S,Takusagawa H,Shen L,et al.Long-term complications associated with glaucoma drainage devices and Boston keratoprosthesis.Am J Ophthalmol.2012;154(1):207-208.[15]Wang L,Jeong KJ,Chiang HH,et al.Hydroxyapatite for keratoprosthesis biointegration.Invest Ophthalmol Vis Sci. 2011;52(10):7392-7399.[16]Kanayama S,Garty S,Kim B,et al.Histological study of graft failure in AlphaCor transplantation.Int Ophthalmol.2011;31(6): 501-504.[17]Liu C,Hille K,Tan D,et al.Keratoprosthesis surgery.Dev Ophtha lmol.2008;41:171-186. [18]Myung D,Duhamel PE,Cochran JR,et al.Development of hydrogel-based keratoprostheses: a materials perspective. Biotechnol Prog.2008;24(3):735-741. [19]Magalhães FP,Sousa LB,Oliveira LA.Boston type I keratoprosthesis: Review.Arq Bras Oftalmol.2012;75(3): 218-222. [20]Harissi-Dagher M,Khan BF,Schaumberg DA,et al.Importance of nutrition to corneal grafts when used as a carrier of the Boston Keratoprosthesis.Cornea. 2007;26(5):564-568. [21]Gomaa A,Comyn O,Liu C.Keratoprostheses in clinical practice-a review.Clin Experiment Ophthalmol.2010;38(2): 211-224. [22]Hicks CR,Hamilton S. Retroprosthetic membranes in Alpha-Cor patients: risk factors and prevention.Cornea.2005; 24(6):692-698. [23]Hicks CR,Vijayasekaran S,Chirila TV,et al.Implantation of PHEMA keratoprostheses after alkali burns in rabbit eyes. Cornea.1998;17:301-308.[24]Lou X,Dalton PD,Chirila TV.Hydrophilic sponges based on 2-hydroxyethyl methacrylate: part VII: modulation of sponge characteristics by changes in reactivity and hydrophilicity of crosslinking agents.J Mater Sci Mater Med.2000;11:319- 325.[25]Vijayasekaran S,Chirila TV,Robertson TA,et al.Calcification of poly(2-hydroxyethyl methacrylate) hydrogel sponges implanted in the rabbit cornea: a 3-month study.J Biomater Sci Polym Edn.2000;11:599-615. |